|
The autophagy inhibitor BCL2 binds to BECN1, reducing its ability to nucleate phagophore formation. Īdditional support for the role of macroautophagy in extending healthy lifespan in mice comes from inbred C57BL/6J mice with a homozygous F121A knock-in mutation to the gene encoding BECN1 ( becn1 F121A/F121A). 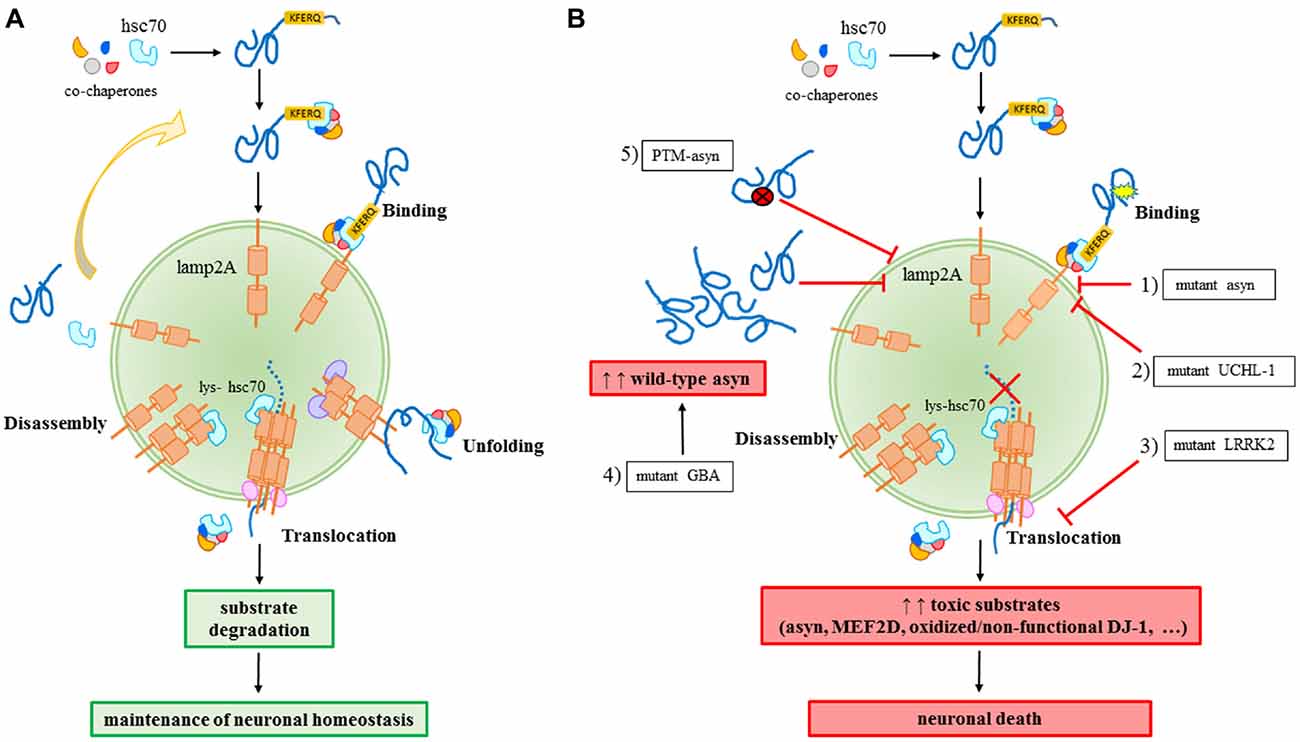
At 6 and 12 months of age, ATG5 TG mice have improved performance on wire hang tests. However, ATG5 TG mice have reduced adiposity, increased insulin sensitivity, increased oxygen consumption rates, and reduced circulating leptin levels. ATG5 TG mice develop normally, show no differences in food intake, and have normal memory (as evaluated by Y-maze tests). showed that two strains of C57BL/6J mice overexpressing ATG5 (ATG5 TG) had a 17% increase in median lifespan, with no sex differences. The first study to show that genetic activation of macroautophagy could extend lifespan in mice was published in 2013, when Pyo et al. However, a few mouse studies do suggest a strong link between MA and longevity. Studies of autophagy and aging in mouse models are not yet as comprehensive as those in invertebrates. Drug treatments or genetic modifications that enhance macroautophagy reliably extend the lifespans of invertebrates. Each branch of autophagy is regulated independently, and each is responsible for degrading a different subset of intracellular cargoes. Lysosomal proteolysis in mammalian cells occurs through at least three distinct pathways: macroautophagy (MA), microautophagy (mA), and chaperone-mediated autophagy (CMA). Protein turnover via the lysosome/autophagy system is a crucial regulator of aging. We found no evidence for age-related changes in LAMP2A levels, CMA substrate uptake, or whole liver levels of CMA degradation targets, despite identifying sex differences in CMA. 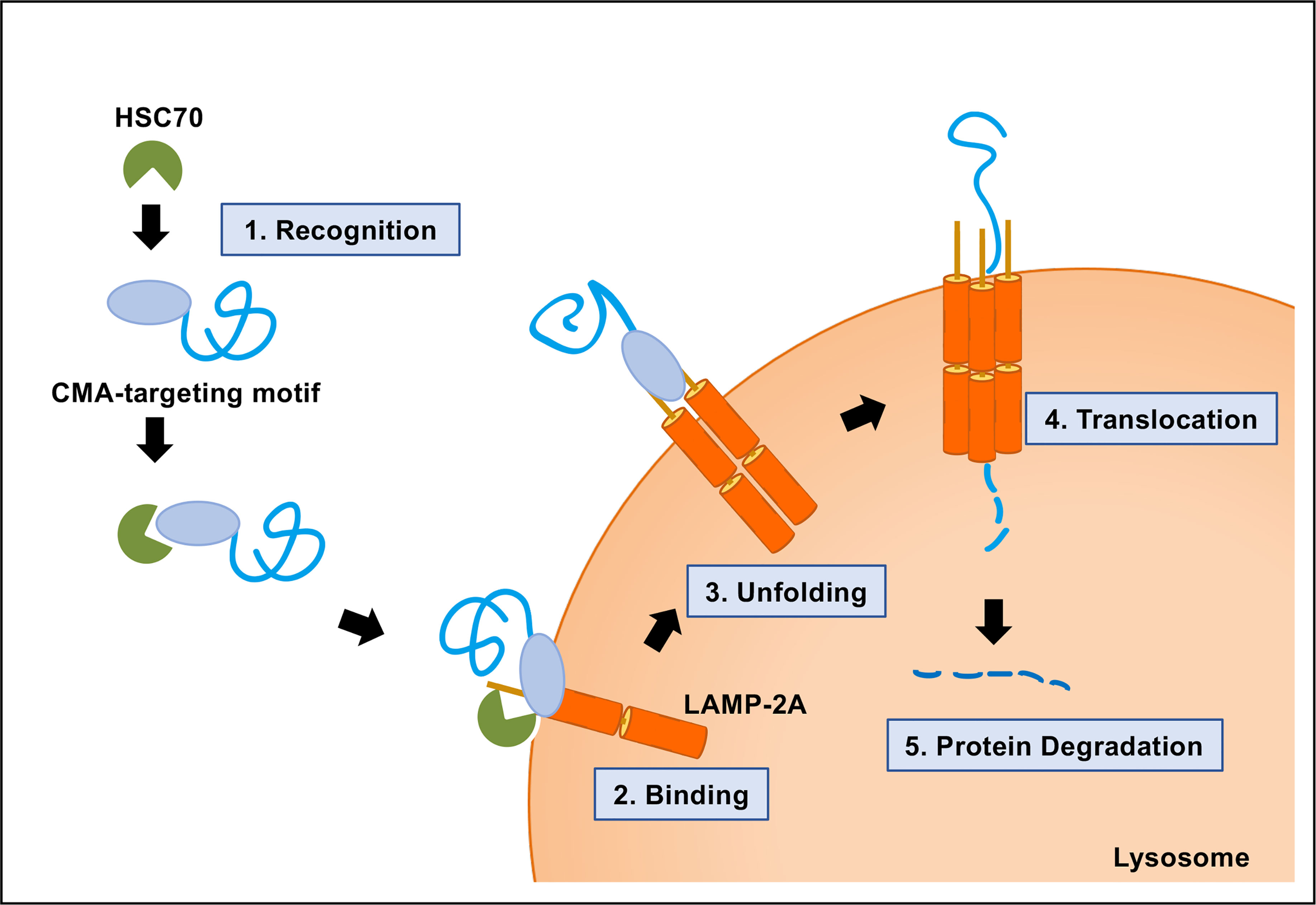
We assessed LAMP2A levels and CMA substrate uptake in both sexes of the genetically heterogeneous UM-HET3 mouse stock, which is the current global standard for the evaluation of anti-aging interventions. This has led to a paradigm in the field of CMA research, stating that the age-associated decline in LAMP2A in turn decreases CMA, contributing to the pathogenesis of late-life disease. Several previous studies, each of which evaluated males of a single inbred mouse or rat strain, have reported that CMA declines with age in many tissues, attributed to an age-related loss of LAMP2A, the primary and indispensable component of the CMA translocation complex. Chaperone-mediated autophagy (CMA) selectively degrades proteins that are crucial for glycolysis, fatty acid metabolism, and the progression of several age-associated diseases.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
